Are babies at greater risk from vaccine than Covid?
Analysing vaccine risk v benefit as approval issued for infants
Irish authorities have issued the seal of approval for the inoculation of infants aged six months, beginning next week.
Children from six months to four years old may be vaccinated against covid 19 beginning Monday, Feb 20.
Immunisation Bulletin 59, issued by the HSE’s National Immunisation Office on February 3 last, deals with new mRNA covid vaccine formulations for children aged six months to four years.
The bulletin notes that the roll out is occurring despite a high prevalence of antibodies and low infection rates.
The latest advice issued in relation to the vaccination of children in the six month to four year age group notes that the need for inoculation in this age group is ‘nuanced’ and should be considered only in line with ‘known risk factor for severe disease.’
“Internationally 50-97% of hospitalised children in this age group had no known risk factor for severe disease,” the bulletin states.
“The benefits of COVID-19 vaccination for the healthy child are more nuanced and are influenced by history and timing of previous infection as well as the need to minimise risks of COVID-19 within a household,” the advice states.
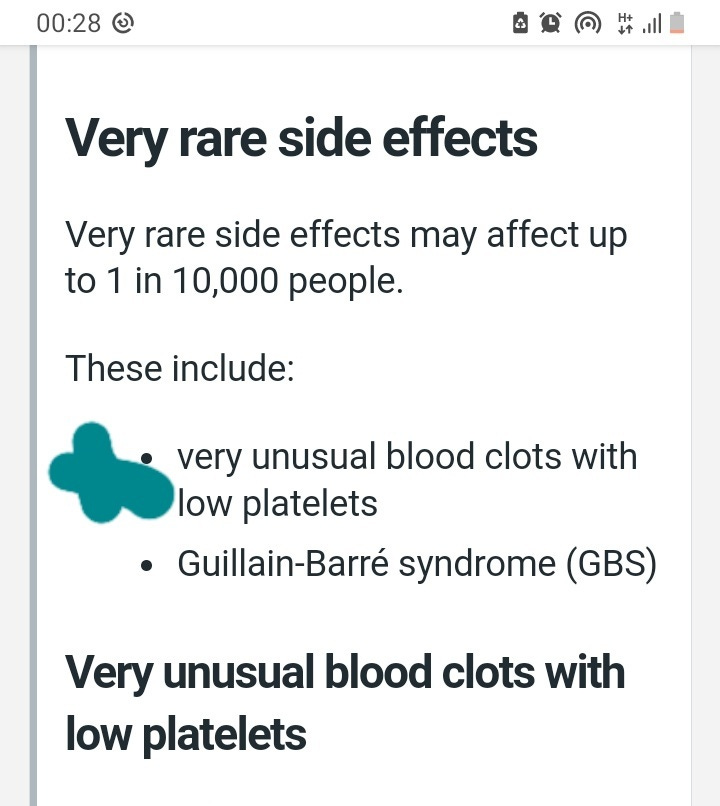
Myocarditis, a potentially fatal condition causing inflammation of the heart, is listed as a side effect that may occur at a rate of one in 10,000. Pericarditis, inflammation of the lining off the heart is also listed as a one in 10,000 risk. (see link, section 4.)
“These conditions can develop within just a few days after vaccination and have primarily occurred within 14 days. They have been observed more often after the second vaccination, and more often in younger males.
“The risk of myocarditis and pericarditis seems lower in children ages 5 to 11 years compared with ages 12 to 17 years. Following vaccination, you should be alert to signs of myocarditis and pericarditis, such as breathlessness, palpitations and chest pain, and seek immediate medical attention should these occur.”
The advice notes that a child who is ‘allergic to the active substance or any of the ingredients of this medicine’ should not receive it.
However, it is not clear how parents or guardians could not know if a child is allergic to the active substances in the vaccine - tozinameran - which is messenger RNA - until after they receive the jab.
(See full ingredients list below)
The HSE Immunisation Bulletin further informs readers that the vaccine may not be effective.
“As with any vaccine, Comirnaty Original/Omicron BA.4-5 may not fully protect all those who receive it and it is not known how long you will be protected. The efficacy of Comirnaty Original/Omicron BA.4-5 may be lower in people who are immunocompromised.”
NIAC is the body responsible for advising the Chief Medical Officer and and the Department of Health on vaccines.
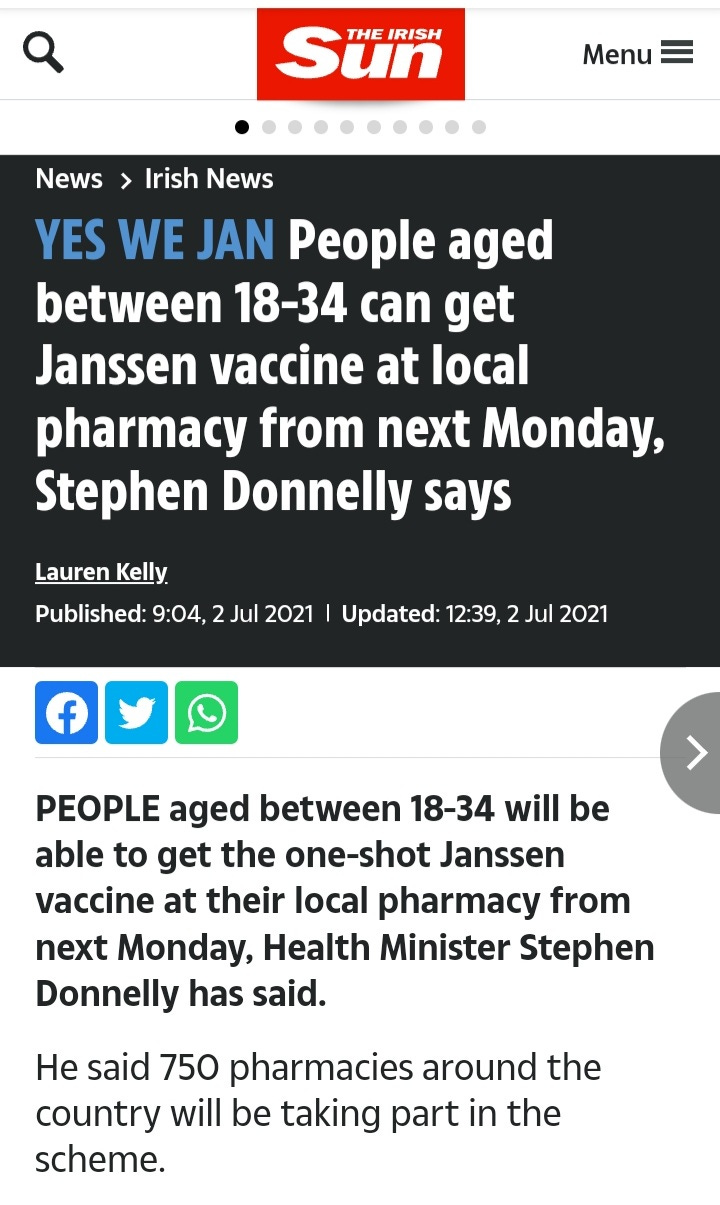
In previous questionable risk benefit analysis, National Immunisation Advisory Committee (NIAC) approved the Janssen vaccine for the 18-34 year age group in June 2021.
Within weeks, the vaccine was quietly withdrawn.
An examination of NIAC advice issued at the time, shows an error in key advice issued.
NIAC Chair Prof Karina Butler told a NPHET briefing the risk of a particular blood clot arising from the Janssen ‘one shot’ vaccine was one in 50,000.
The actual risk of a blood clot with low platelets, according to the product safety leaflet, was one in 10,000. Prof Butler made the comments at a NPHET briefing on July 14 2021. The Janssen vaccine was withdrawn in August.
At the NPHET meeting on July 14 2021, Prof Butler acknowledged that mRNA vaccines (Pfizer and Moderna) were the preferred option for younger age cohorts, but said rising infection rates tipped the balance in favour of the use of Janssen and Astra Zeneca vaccines.
“We have said before that mRNA vaccines are preferred for those age groups and that was because of the very small albeit, but potential risk of the clotting with low platelet counts, the thrombosis thrombocytopenia syndrome that might occur at the rate of about one in 50,000 in those age groups.
“That was the reason in the beginning that there was a preference given for the RNA vaccines because it was balancing that small risk against what was the risk of someone ending up in ICU or seriously ill with covid,” Prof Butler said.
However the Janssen patient safety leaflet placed the risk at one in 10,000, as does the current HSE safety information.
This apparent anomaly was put to press departments at the HSE and to the Pharmaceutical Society of Ireland (PSI) by the author of this Substack in July 2021.

The HSE issued no response, while the PSI said risk factors provided for in patient information leaflets were a matter for the Health Products Regulatory Authority (HPRA).
“The PSI is not involved in the detailed management of the vaccination programme in pharmacies and operational details are the responsibility of the HSE. The PSI also has no role in relation to the specific vaccines and any risk factors provided for in patient information leaflets, this is a matter for the Health Products Regulatory Authority,” a PSI spokesperson said.
Meanwhile, also in July 2021, media company Kinzen, contracted by the HSE, incorrectly flagged social media posts linking Janssen vaccine to blood clots as 'misinformation.'
*
Warm thanks to those that continue to support my work, it is much appreciated.
*The active substances of COVID-19 mRNA Vaccine are tozinameran and famtozinameran.
After dilution, the vial contains 10 doses of 0.2 mL with 5 micrograms to tozinameran (Original) and 5 micrograms of famtozinameran (Omicron BA.4-5) per dose.
The other ingredients are: − ((4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate) (ALC-0315) − 2-[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide (ALC-0159) − 1,2-Distearoyl-sn-glycero-3-phosphocholine (DSPC) − cholesterol − trometamol − trometamol hydrochloride − sucrose − water for injections
Pfizer /Cominarty Contents List - see section 6.







I got GBS Dec 2020. Its no joke to be paralysed from head down. To learn how to hold objects again, put on a ventilator, traceostomy, etc. Expected to die. And I hadn't been vaccinated, through choice (obviously). Yet whilst I was in hospital - the consultant pressured me to take Vax when they were rolled out (circa April/May 2021). So they wanted me, with a totally broken immune system (a immune system attacking its own defensive mylene sheath : GBS) - to take an experimental vax (where 1 in 10,000 would be affected by it as per the withdrawn vax). When I got GBS in Dec 2020, the percentile of contacting it, was one in a million it was very rare (lucky me!!). I got it from trauma. Body shuts down via the central nervous system. When I left hospital (8 months later, i.e. July 2021), the rate of GBS had heightened exponentially - 6 new patients had joined me, whereas when I arrived in Dec 2020, they hadn't had a GBS case in two years. No so rare, afterall, These Days! And now, they are pushing vax on babies, toddlers with an immune system far superior to any other. Apart from the stress of being born, what trauma has a baby had in its life to date?, yet with the huge GBS increase in society since the vax rollout, who would want a young child to contact that?! What cognitive 'fight-to-live' could a child that young have? It took me every fibre of my being to get through it. We are dealing with psychopaths here.
Never was the phrase 'caveat emptor' more relevant.
Thanks to this great piece of investigative journalism we learn that those pushing these things are smart enough to include warnings about myocarditis, etc. in their literature. Perhaps they believe that hedging their bets like this will get them off the hook later on. But, if and when they appear before a human court, that is unlikely.
What they should really be worried about though is what it's doing to them inside. Evil like this corrodes the very soul. Oscar Wilde understood this when he wrote The Picture of Dorian Gray.